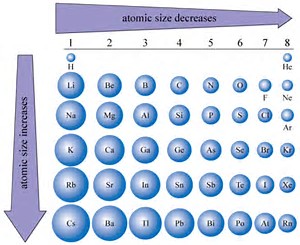
Helium turns out to be the smallest atom on that graph, which is one of the reasons it was discovered in the sun before it was discovered on earth. How does the atomic radius change as the period progresses from left to right As you move from. Whenever an electron shell is filled, adding another electron will cause a step-change in the atomic radius as can be seen in the jagged graph in one of the answers above. Lithium is the largest atom, and oxygen is the smallest atom. You can in principle continue this procedure, whereby each additional proton contracts the radius, and each additional electron expands it slightly. Imagine you start with one proton and one electron this is the $\ce.$ To gain some intuition as to how this works, it may be helpful to do a Gedankenexperiment wherein we build atoms by adding protons and electrons one at a time, neglecting the neutrons which are not important here. Which effect dominates depends on whether or not an additional electron finds a place in an existing electron shell.

Your intuition about more electrons leading to a larger atom is correct, but more protons actually lead to a smaller atom. The size of the atom is therefore only defined by the electron shells (orbitals), which in turn depend on the nuclear charge. Rutherford's gold foil experiment taught us that the protons and neutrons fill only a tiny portion of the atom, which we call the nucleus. muons are heavier so are a lot closer to the nucleus so the atoms are a lot smaller, they are called Muonic atoms
If you do this for hydrogen and helium you get the figure below, where the solid lines are the probability distribution and the dashed lines are the average radius, which show that on average the electrons in helium are closer to the nucleus than in hydrogen.Īlso if you want to break from the periodic table, there are atoms where the electrons are replaced by their subatomic cousins, muons. a more accurate modle for electrons is using quantum physics, in the is system the electron aren't at a specific spot, instead they are described by a quantity called the wave function which gives the probability of the electron being in a specific position.įrom statistics you can then work out the average position (the expectation value), this is the product of the position and how likely it is to be there (the probability) The behaviour of electrons in atoms is not well describes by the classical points and orbits model of the atom.
